Planet Earth—a mere speck in our universe—is residence to extra viruses than there are stars within the cosmos. They exist at life’s outer restrict, performing a fragile dance that has lasted eons however can flip right away. Not alive, but entwined with the dwelling, viruses proceed to problem and shock us.
Like when the information broke in March 2024 that fowl flu had unfold to dairy cows.
“It was exhausting to consider,” stated evolutionary biologist Yvonne Su, who’s an affiliate professor with Duke-NUS’ Emerging Infectious Diseases Programme. Traditionally, cows had been thought to not be vulnerable to fowl flu or influenza A, the kind of influenza that circulates freely in wild water and shorebirds.
Initially startled, her instant subsequent thought had been, “Is there direct cow-to-cow transmission?”
When we spoke in early June, Su talked me by what little knowledge was out there at the moment. While the variety of unanswered questions rivaled the variety of herds contaminated, some issues have been starting to emerge.
“We realize it binds to the mammary glands in cows and we are able to see some cow-to-cow transmission,” stated Su.
By the tip of the month, the genetic tree tracing the virus’ evolution revealed an explosive epidemic. Virologist Gavin Smith, who leads Duke-NUS’ Emerging Infectious Diseases Programme, summed up what he noticed:
“Everything may be very carefully associated to one another. If you have a look at the distribution, it comes from a background the place this virus may be very widespread in wild birds and the atmosphere.”
By mid-July, the virus had contaminated cows on 161 dairy farms in 13 US States, together with thousands and thousands of poultry throughout 37 farms and 9 people.
And that was within the US alone. In Southeast Asia, H5N1—albeit a genetically completely different variant—has undergone a resurgence in poultry and is understood to have contaminated not less than 13 people in Cambodia since 2023, with further instances reported in Vietnam and China.
“The current surge in avian influenza outbreaks is deeply regarding,” stated Kachen Wongsathapornchai, Regional Manager of the Food & Agriculture Organization’s Emergency Centre for Transboundary Animal Diseases, in a press release.
“Since late 2023, we’ve got noticed an increase in human instances and the virus spreading to new animal species. The emergence of novel A/H5N1 strains, that are extra simply transmissible, will increase the pandemic menace. Immediate, coordinated preventive measures are important.”
How to construct a pandemic
Influenza viruses have co-existed with people since as early because the sixteenth century. Easily transmissible and extremely infectious, they flow into the globe freely.
“It is estimated that by the age of 5, most individuals could have been contaminated with influenza,” stated Smith.
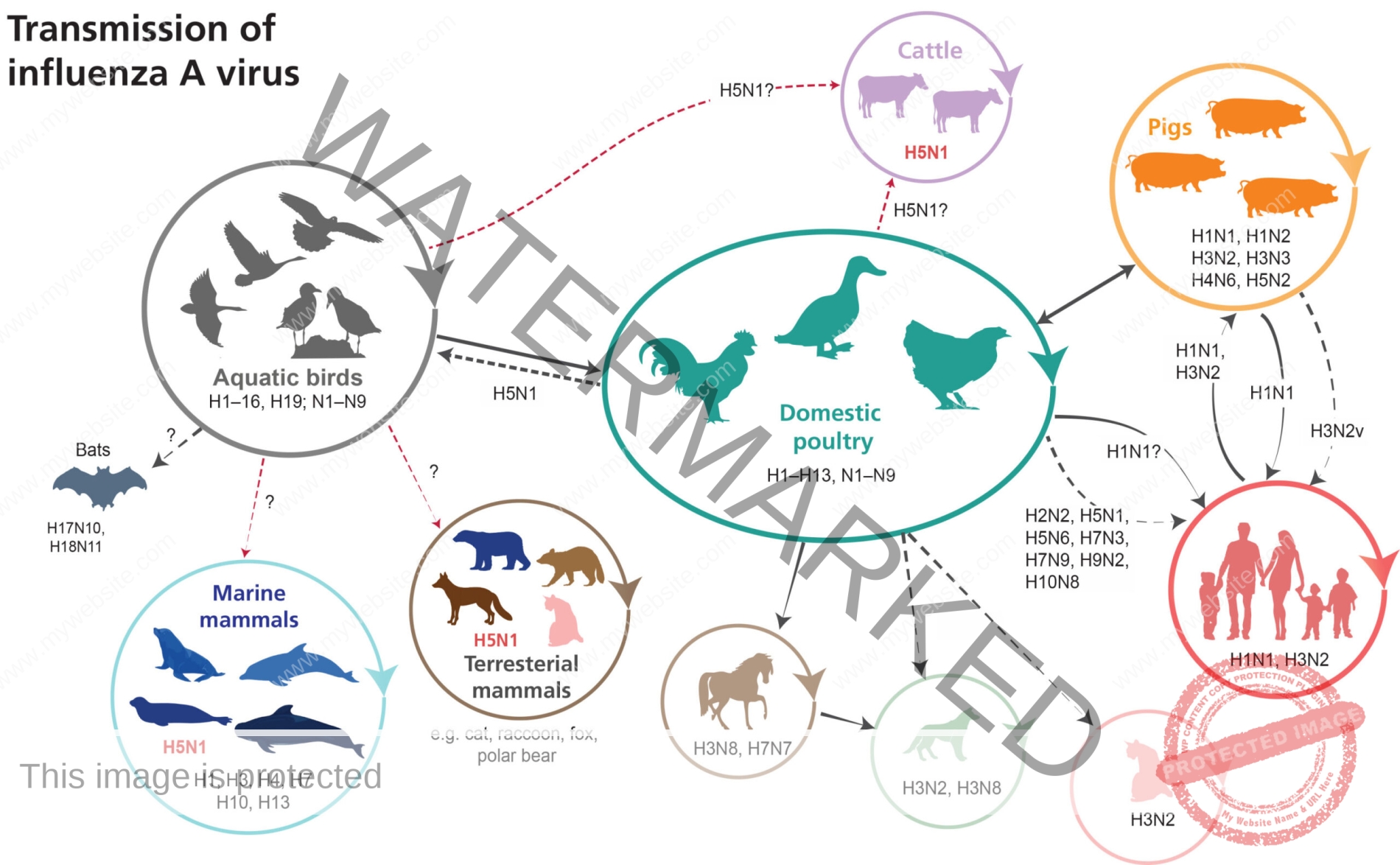
And it isn’t just some kinds of animals which can be contaminated with the virus. While influenza viruses fall into 4 varieties, it’s actually influenza A that has the widest attain, infecting nearly all types of life—from birds to polar bears, from seals to, now, cows.
And each one of many 14 influenza pandemics in people is squarely the results of an influenza A virus.
While H5N1 hasn’t triggered an outbreak in people on the dimensions of H1N1, variations of which have been liable for each the 1918 and 2009 pandemics, it does maintain an ignoble honor: it’s the first confirmed fowl flu that efficiently jumped instantly from birds to people.
“I used to be informed that that outbreak was a complete shock,” stated Smith of the 1997 H5N1 outbreak in Hong Kong that killed one out of three folks contaminated with the virus. Their blood had thickened to treacle, their lungs, livers and kidneys failing one after the other like a line of dominoes. All had been in shut contact with sick chickens.
“Before that, it was thought that the virus wanted to come back from a mammalian middleman host,” added Smith, who has been engaged on influenza for greater than twenty years.
An middleman host, similar to pigs, whose respiratory tracts are lined with docking stations, or receptors, for each avian influenza and human influenza.
“That is why they’re such good mixing vessels. They could be contaminated by a human influenza virus and fowl virus on the identical time, and these viruses can reassort their genes,” stated Su. “That’s how we acquired the H1N1 pandemic virus in 2009.”
But that did not occur in 1997. The outbreak ended after a four-day-long slaughter, the place all 1.6 million chickens within the territory have been culled. As a precaution, stay poultry markets have been shuttered for seven weeks. Trade stood nonetheless and the world held its breath. But it was sufficient.
“The virus that contaminated folks in Hong Kong has died out and by no means been detected in poultry once more,” stated Smith.
But its siblings, tracked by the shared HA or hemagglutinin that they keep on the skin, continued.
“The hemagglutinin is what binds to the host receptor, making it much like the SARS-CoV-2 spike protein,” stated Su.
After mendacity low till the flip of the century, H5N1 re-emerged throughout Asia, inflicting widespread outbreaks in poultry, earlier than spilling again into wild birds in 2005.
And it was that transmission again into wild water birds that helped H5N1 escape out of Asia and set up itself in poultry in Europe and Africa.
“But it did not appear very nicely established in wild birds,” stated Smith.
As the virus mingled with different strains that have been circulating in Europe and Africa, it adopted the trail of survival mindlessly, sampling from an enormous array of attainable new combos of inside in addition to exterior ones, together with with the numerous completely different neuraminidases, or Ns. These fellow floor proteins launch the virus out of the contaminated cells as soon as it has made sufficient copies of itself.
With a wealth of 18 Hs and 11 Ns to select from, on high of evolutionary mutations, influenza is nearly as advanced as the newest algorithm underpinning massive language fashions.
“Besides the truth that you might have the range within the H and the N, the influenza virus is completely different from the SARS coronavirus in that its genome is in eight separate strands of RNA,” stated clinician-scientist Ooi Eng Eong, a professor with Duke-NUS’ Emerging Infectious Diseases Programme.
“Now if two viruses, say H5N1 and H3N2, go into the identical animal, then you may get all kinds of blending. Out can come a virus that’s H5N2, or H3N1, and the interior genes can be combined. It is that this mixing that then opens a number of doorways for the virus to contaminate new animals.”
This is exactly what occurred. Between 2016 to 2019, H5 diversified, combining with N2, N6 and N8, till the H5N8 pressure gained some dominance in Asia and Europe, inflicting massive outbreaks in poultry.
Then in 2020, the H5 virus rejoined with an N1 gene, this time one which had developed in wild birds.
“Suddenly, we’ve got a virus that may be very nicely tailored to wild geese. And it is that virus that acquired into the US and South America,” stated Smith.
The virus was not solely nicely tailored, but it surely additionally unfold explosively. And it’s lethal.
When Su lined up the hemagglutinin gene sequences from the newest descendants on this outbreak, she pointed to a selected grouping of nucleotides across the 1,000-base mark that was affected by Rs and Ks, marking them as viruses that may trigger extreme illness.
“When we see this sample, we all know this virus has a polybasic cleavage web site, which implies it’s a extremely pathogenic sort of influenza,” stated Su.
In poultry like chickens, influenza viruses with polybasic cleavage websites typically infect cells extra simply, inflicting widespread harm. Wild water birds, like geese, undergo solely transient or no signs in any respect.
“Unfortunately for us, extremely pathogenic influenza in chickens occurs to even be fairly lethal in people,” stated Smith.
And whereas the pressure from 1997 has not been seen since, sporadic instances of human H5N1 infections proceed to happen, with 889 instances reported to the World Health Organization between 1 January 2003 and three May 2024. Its fatality charge? More than 50%.
‘Teatering’ on the tip of the subsequent pandemic?
For now, the pressure of H5N1 detected within the US has not made its manner again to Asia. But this might simply be a matter of time. Apart from geographic unfold, virologists and public well being officers are additionally on alert for different hazard indicators.
“The receptor binding profile. If that modifications, that may be a concern,” stated Smith, as a result of for now, H5N1 remains to be essentially a fowl flu. But with the viruses replicating exponentially, it would not take a greater tailored variant lengthy to muscle out its weaker brethren.
“Another concern is that it acquires some particular mutations within the polymerase genes which can be related to improved replication in people,” added Smith.
Unlike the 2 floor genes, the polymerase genes are tucked away contained in the virus. While they do not have an effect on the virus’ means to bind to cells or to contaminate them, they decide how nicely the virus can copy itself. Even if a person is contaminated with a virus whose polymerase genes do not carry that mutation, by the tip of their an infection, the mutation extra probably than to not have developed spontaneously.
“So if these viruses come pre-loaded with this mutation that may be a little bit of a priority,” added Smith.
Widespread infections amongst pigs are one other concern as a result of to this point H5N1 appears to have shunned pigs. In reality, all through the a long time of circulating between birds and poultry and occasionally into people, H5N1 appears to have struggled to duplicate in pigs.
Of course, to this point pigs—and cows—have solely had their noses swabbed for virus samples.
“All the earlier surveillance on the lookout for infections targeted on the respiratory tract. No one checked out what occurs within the teats,” stated Smith.
Even although, Smith was fast to level out, scientists from Canda’s Department of Agriculture had demonstrated {that a} sort of human influenza A might infect the mammary glands of cows again in 1953.
“Why they began within the largest animal, nobody is aware of, but it surely’s humorous how rapidly we overlook,” he added.
When requested whether or not this may very well be the start of the subsequent pandemic, Su whispered “I hope not,” as if her phrases might develop into a self-fulfilling prophecy if uttered too loud.
To do her half, Su goes again over all her samples collected not simply from pigs, however goats and sheep too, testing them for the shadow presence of a virus as soon as alive. Particularly in pigs, these viruses are recognized to persist for many years, all the time lurking within the background able to reassort with different viruses. But she’s additionally eager about the implications of this outbreak on future work.
“We want to consider altering the place we swab completely different animals after we do surveillance. Because the route of transmission might be key to serving to us perceive not simply how an animal acquired contaminated however how we are able to management an outbreak and forestall it from escalating.”
How cows within the US acquired contaminated stays an unanswered query. The newest proof, printed by Nature on 25 July by way of early entry, leaves room for a respiratory route in addition to direct an infection of the mammary gland by contaminated flooring and bedding, or mechanically by way of milking tools.
Expanding these early observations Su made in June, the researchers noticed that their investigation “supplies proof of environment friendly intra- and inter-species transmission.”
And if this virus is not simply nicely tailored to cows however spills freely from cows again into poultry and from there again into wild birds or establishes itself in pigs, it’s anybody’s guess what is going to come out.
Feeding the subsequent pandemic
Such rampant unfold by the meals manufacturing system is what makes the present scenario so perilous.
“The extra components of your meals manufacturing system that you’ve got contaminated and the extra people who find themselves presumably uncovered, the higher the danger,” stated Smith. “Regardless of whether or not that may be a danger of reassortment or an adaptation, that state of affairs supplies extra alternative for both of these.”
And as a result of the variety of particular person virus particles is so excessive throughout an an infection, inhabitants stage modifications can happen inside a single replication spherical.
“So, if you’re contaminated with viruses, the place 20% bind to human receptors, and 80% bind to fowl receptors, within the subsequent spherical, you’ll solely have viruses that bind to the human receptor,” stated Smith.
Catching the virus earlier than this occurs is inconceivable. The neatest thing that may be carried out is to scale back the quantity of viruses on the market by culling of contaminated animals and vaccinating at-risk populations. Vaccinating high-risk farm staff and updating the newest pandemic vaccine strains that must be on standby are different precautions.
For Renzo Guinto, an affiliate professor of Global and Planetary Health with the SingHealth Duke-NUS Global Health Institute, this is a chance to strike. “Since zoonotic outbreaks push us to seek out methods to enhance an infection security in the way in which we produce our meals, notably animal meat merchandise, we’ve got a chance to remodel meals methods extra comprehensively—to make them environmentally sustainable, resilient to shocks and accessible to all.”
With the current resurgence of various H5N1s world wide, the prospect of a variant that’s match to unfold in people freely stays a continuing menace.
But no matter how occasions unfold on a human stage, H5N1 has already wrought untold harm.
“We have seen massive die-offs amongst many species, it is spilled into marine mammals, it makes pigs sick, wreaks havoc in poultry manufacturing and now it impacts cow’s milk manufacturing,” enumerates Smith. “It is an ecological catastrophe.”
An ecological catastrophe with humanity’s fingerprints throughout it. So understanding this interface, the place people, farmed animals and wild birds collide, might be important as we search to hint the evolution and unfold of H5N1. And in doing so, we may uncover tips on how to fortify life’s borders in opposition to these invisible threats.
Duke-NUS Medical School
Citation:
The subsequent pandemic? How a well-known virus exploits new hosts (2024, August 27)
retrieved 27 August 2024
from
This doc is topic to copyright. Apart from any truthful dealing for the aim of personal examine or analysis, no
half could also be reproduced with out the written permission. The content material is supplied for info functions solely.